Do Metals Form Cations
Do Metals Form Cations - Web alkali metals and alkaline earth metals always form cations. Oxygen, nitrogen, sulfur), while most metals form cations (e.g. What types of ions do the metallic and the nonmetallic elements form? And because of this behavior,. Cl atoms gain 1 electron, therefore making them anions. You can also tell that they form cations because some. They are formed when a metal loses its electrons. Therefore, they possess a net positive charge. Web having lost electrons, which are negatively charged, atoms of metals therefore gain a net positive charge i.e they become cations. Web halogens always form anions, alkali metals and alkaline earth metals always form cations.
They are formed when a metal loses its electrons. Most other nonmetals typically form anions (e.g. What types of ions do the metallic and the nonmetallic elements form? Web alkali metals and alkaline earth metals always form cations. Web a metal ion in aqueous solution or aqua ion is a cation, dissolved in water, of chemical formula [m(h 2 o) n] z+. Writing chemical formulas when writing the formula of a compound, the cation is listed before the anion. Web cations are positively charged ions. Oxygen, nitrogen, sulfur), while most metals form cations (e.g. Pearson higher level chemistry textbook, 2nd edition. And because of this behavior,.
Web having lost electrons, which are negatively charged, atoms of metals therefore gain a net positive charge i.e they become cations. Cl atoms gain 1 electron, therefore making them anions. All chemical reactions are driven to gain more. And because of this behavior,. It has fewer electrons than protons. Web cations are atoms that contain a positive charge, and they are formed when the atoms lose electrons which are negatively charged. This is the typical behavior for many metal substances. Closely packed cations and loosely held valence electrons rather than neutral atoms what can the valence electrons in a pure metal be modeled as? Pearson higher level chemistry textbook, 2nd edition. Therefore, they possess a net positive charge.
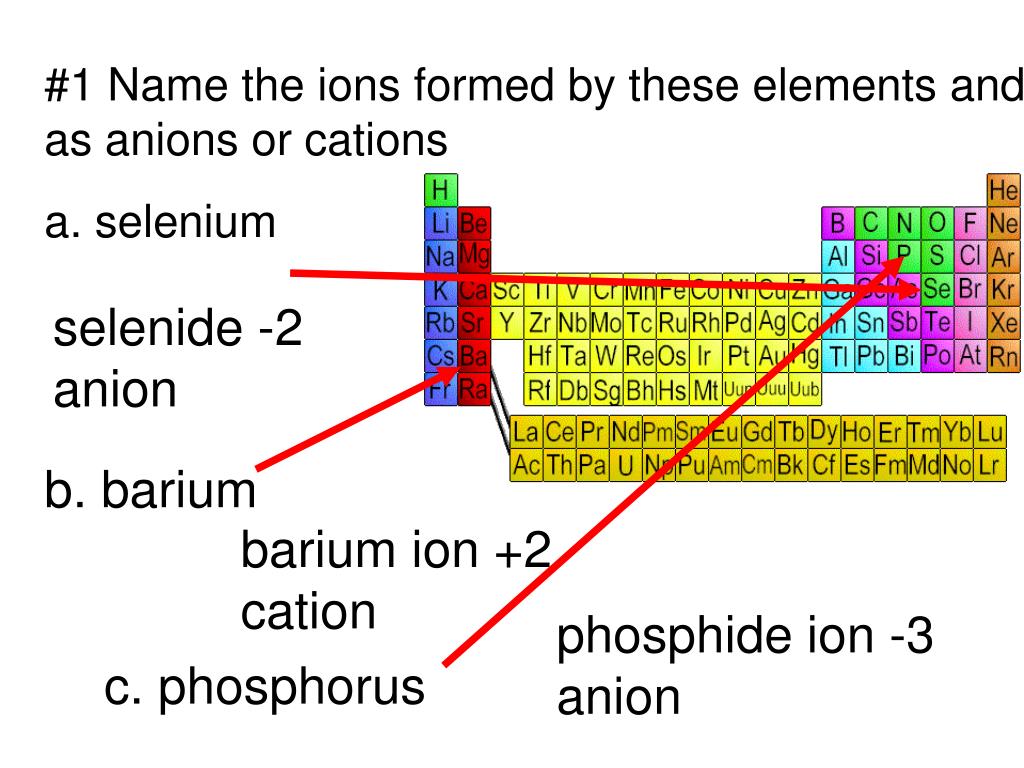
PPT 1 Name the ions formed by these elements and classify them as
Therefore, they possess a net positive charge. What types of ions do the metallic and the nonmetallic elements form? Web cations are positively charged ions. Web halogens always form anions, alkali metals and alkaline earth metals always form cations. Web having lost electrons, which are negatively charged, atoms of metals therefore gain a net positive charge i.e they become cations.
*2.7 Nomenclature Chemistry LibreTexts
Web metals tend to form cations while nonmetals tend to form anions. Web halogens always form anions, alkali metals and alkaline earth metals always form cations. What types of ions do the metallic and the nonmetallic elements form? Web cations are atoms that contain a positive charge, and they are formed when the atoms lose electrons which are negatively charged..
is incorreer? all s area eleseis erand e are metals b, all p area
Web a metal ion in aqueous solution or aqua ion is a cation, dissolved in water, of chemical formula [m(h 2 o) n] z+. Web halogens always form anions, alkali metals and alkaline earth metals always form cations. Web having lost electrons, which are negatively charged, atoms of metals therefore gain a net positive charge i.e they become cations. Pearson.
PPT Chapter 9 The Basics of Chemical Bonding PowerPoint Presentation
All chemical reactions are driven to gain more. They lose one or more than one electron. You can also tell that they form cations because some. Cl atoms gain 1 electron, therefore making them anions. This is the typical behavior for many metal substances.
PPT Naming IONS & formulas for Ionic Compounds PowerPoint
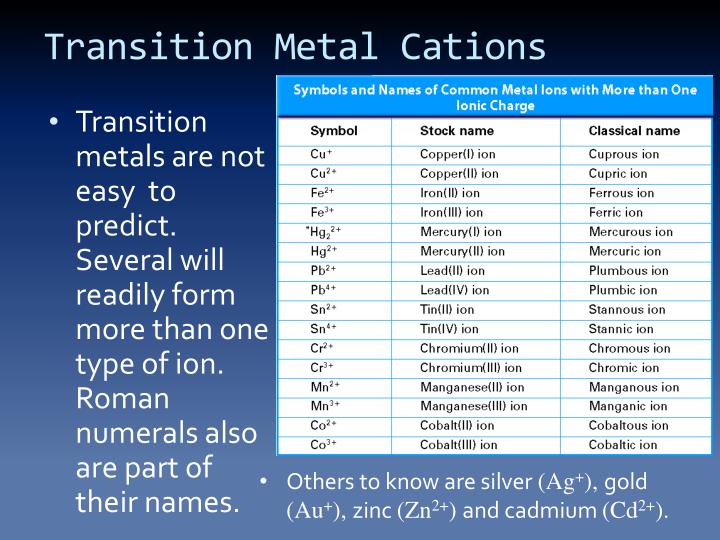
All chemical reactions are driven to gain more. Web cations are positively charged ions. Transition metals often form ions wihout complete octets that's why all the stable ions are all cations. The solvation number , n , determined by a variety of experimental methods is 4 for li + and be 2+ and 6 for most elements in periods 3..
Do metals form anions or cations quizlet? Book Vea
Web cations are atoms that contain a positive charge, and they are formed when the atoms lose electrons which are negatively charged. Web alkali metals and alkaline earth metals always form cations. They lose one or more than one electron. Web cations are positively charged ions. Web what do metals consist of?
Chem matters ch6_ionic_bond
This is the typical behavior for many metal substances. It has fewer electrons than protons. All chemical reactions are driven to gain more. What types of ions do the metallic and the nonmetallic elements form? Cl atoms gain 1 electron, therefore making them anions.
Solved Question 6 (1 point) Why do metals tend to form
Web a metal ion in aqueous solution or aqua ion is a cation, dissolved in water, of chemical formula [m(h 2 o) n] z+. Web having lost electrons, which are negatively charged, atoms of metals therefore gain a net positive charge i.e they become cations. Oxygen, nitrogen, sulfur), while most metals form cations (e.g. This is the typical behavior for.
PPT IONS PowerPoint Presentation ID2435906
By catrin brown and mike ford. They are formed when a metal loses its electrons. You can also tell that they form cations because some. Therefore, they possess a net positive charge. Web cations are atoms that contain a positive charge, and they are formed when the atoms lose electrons which are negatively charged.
Form a table of alkaline earth metal cations and the
Web cations are positively charged ions. Closely packed cations and loosely held valence electrons rather than neutral atoms what can the valence electrons in a pure metal be modeled as? Therefore, they possess a net positive charge. And because of this behavior,. Cl atoms gain 1 electron, therefore making them anions.
By Catrin Brown And Mike Ford.
It has fewer electrons than protons. All chemical reactions are driven to gain more. This is the typical behavior for many metal substances. They are formed when a metal loses its electrons.
Web Cations Are Positively Charged Ions.
Web having lost electrons, which are negatively charged, atoms of metals therefore gain a net positive charge i.e they become cations. Therefore, they possess a net positive charge. Pearson higher level chemistry textbook, 2nd edition. Transition metals often form ions wihout complete octets that's why all the stable ions are all cations.
Web What Do Metals Consist Of?
And because of this behavior,. Web a metal ion in aqueous solution or aqua ion is a cation, dissolved in water, of chemical formula [m(h 2 o) n] z+. Web alkali metals and alkaline earth metals always form cations. The solvation number , n , determined by a variety of experimental methods is 4 for li + and be 2+ and 6 for most elements in periods 3.
Most Other Nonmetals Typically Form Anions (E.g.
You can also tell that they form cations because some. Writing chemical formulas when writing the formula of a compound, the cation is listed before the anion. Closely packed cations and loosely held valence electrons rather than neutral atoms what can the valence electrons in a pure metal be modeled as? What types of ions do the metallic and the nonmetallic elements form?