Molnupiravir Patient Fact Sheet
Molnupiravir Patient Fact Sheet - Food and drug administration (fda) has issued an eua for the emergency use of the unapproved. Web the molnupiravir (mov) eua factsheets are being revised at this time for the following two reasons: Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for. Red, swollen, blistered, or peeling skin with or without fever; To account for the fda. Web signs of an allergic reaction, like rash; Pediatric (for additional information see molnupiravir (united states: Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for.
Pediatric (for additional information see molnupiravir (united states: Web the molnupiravir (mov) eua factsheets are being revised at this time for the following two reasons: Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for. Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for. To account for the fda. Web signs of an allergic reaction, like rash; Red, swollen, blistered, or peeling skin with or without fever; Food and drug administration (fda) has issued an eua for the emergency use of the unapproved.
Pediatric (for additional information see molnupiravir (united states: To account for the fda. Food and drug administration (fda) has issued an eua for the emergency use of the unapproved. Red, swollen, blistered, or peeling skin with or without fever; Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for. Web the molnupiravir (mov) eua factsheets are being revised at this time for the following two reasons: Web signs of an allergic reaction, like rash; Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for.
Molnupiravir (new antiviral) Interim Data Unambiguous Science
Web the molnupiravir (mov) eua factsheets are being revised at this time for the following two reasons: Food and drug administration (fda) has issued an eua for the emergency use of the unapproved. Web signs of an allergic reaction, like rash; Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for. Web fact sheet.
A Comparison Of Paxlovid Versus Molnupiravir The First Oral COVID
Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for. Food and drug administration (fda) has issued an eua for the emergency use of the unapproved. Web the molnupiravir (mov) eua factsheets are being revised at this time for the following two reasons: Pediatric (for additional information see molnupiravir (united states: Web signs of.
Merck’s Molnupiravir Second COVID Pill Is SecondLine, But Timing
Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for. Red, swollen, blistered, or peeling skin with or without fever; Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for. Web the molnupiravir (mov) eua factsheets are being revised at this time for the following two reasons: Pediatric.
Patient Handouts — TeamTAUC
Red, swollen, blistered, or peeling skin with or without fever; Web the molnupiravir (mov) eua factsheets are being revised at this time for the following two reasons: Web signs of an allergic reaction, like rash; Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for. Pediatric (for additional information see molnupiravir (united states:
Test and Treat COVID19 Community Guide to Molnupiravir Treatment
Web signs of an allergic reaction, like rash; Food and drug administration (fda) has issued an eua for the emergency use of the unapproved. Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for. Red, swollen, blistered, or peeling skin with or without fever; Pediatric (for additional information see molnupiravir (united states:
Today MOLNUPIRAVIR Medication News, Jan 28, 2022
Food and drug administration (fda) has issued an eua for the emergency use of the unapproved. Web the molnupiravir (mov) eua factsheets are being revised at this time for the following two reasons: Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for. Red, swollen, blistered, or peeling skin with or without fever; Web.
Molnupiravir Fact Sheet for Healthcare Givers and Recipients
Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for. Web signs of an allergic reaction, like rash; Pediatric (for additional information see molnupiravir (united states: To account for the fda. Web the molnupiravir (mov) eua factsheets are being revised at this time for the following two reasons:
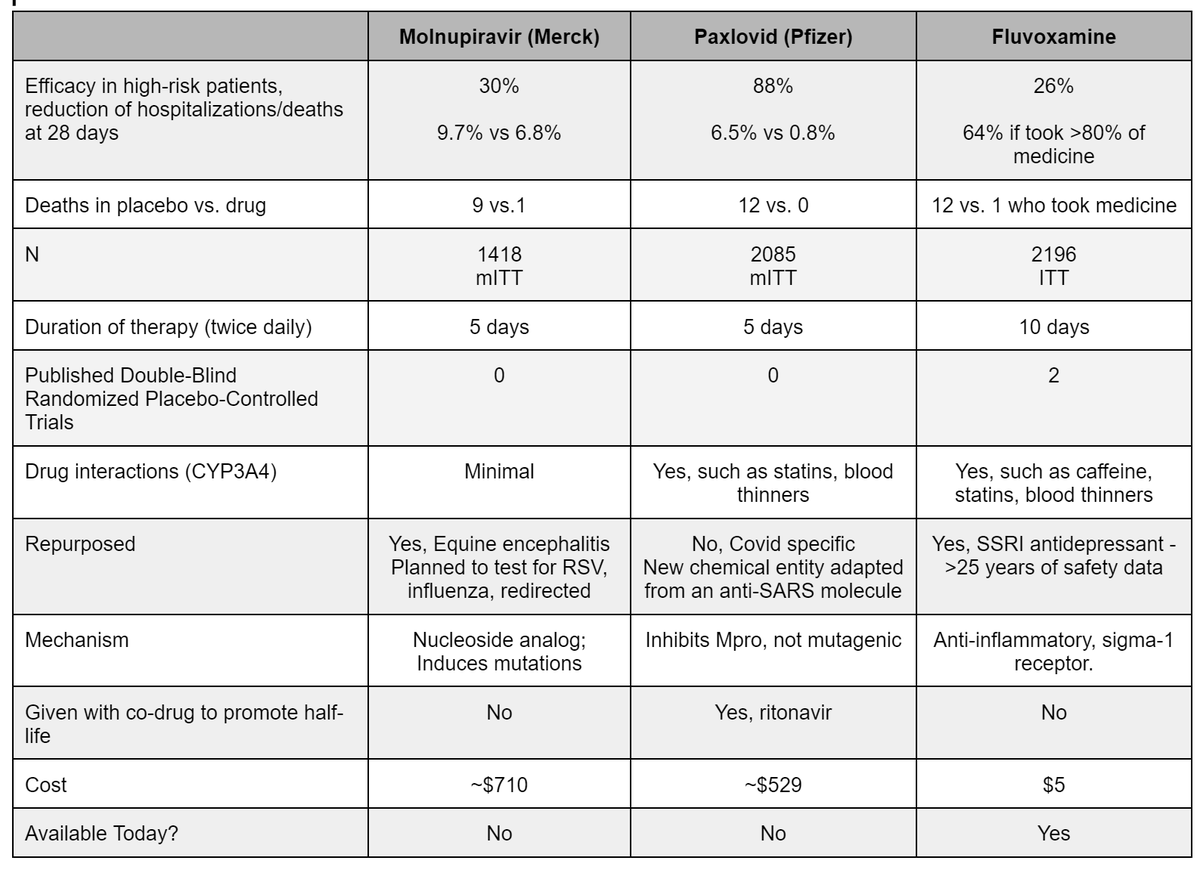
Updated chart on early COVID19 treatment for Molnupiravir,
Pediatric (for additional information see molnupiravir (united states: Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for. Red, swollen, blistered, or peeling skin with or without fever; Web the molnupiravir (mov) eua factsheets are being revised at this time for the following two reasons: Web fact sheet for patients and caregivers emergency use.
Molnupiravir STELLA 400 mg GONSA
To account for the fda. Pediatric (for additional information see molnupiravir (united states: Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for. Food and drug administration (fda) has issued an eua for the emergency use of the unapproved. Red, swollen, blistered, or peeling skin with or without fever;
Medicines factsheets Healthify
Web the molnupiravir (mov) eua factsheets are being revised at this time for the following two reasons: Food and drug administration (fda) has issued an eua for the emergency use of the unapproved. Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for. Red, swollen, blistered, or peeling skin with or without fever; Web.
Web The Molnupiravir (Mov) Eua Factsheets Are Being Revised At This Time For The Following Two Reasons:
Web signs of an allergic reaction, like rash; Food and drug administration (fda) has issued an eua for the emergency use of the unapproved. Red, swollen, blistered, or peeling skin with or without fever; Pediatric (for additional information see molnupiravir (united states:
Web Fact Sheet For Patients And Caregivers Emergency Use Authorization (Eua) Of Lagevriotm (Molnupiravir) Capsules For.
To account for the fda. Web fact sheet for patients and caregivers emergency use authorization (eua) of lagevriotm (molnupiravir) capsules for.